.jpg)
.jpg)
Miltenyi Bioindustry offers advanced cell therapy manufacturing services tailored to the needs of preclinical and early clinical biopharma programs. With global GMP-compliant facilities and decades of experience, we support the entire development journey – from early process development to commercial-scale production. Our expertise, proprietary technologies, and IND-readiness strategies offer a faster, more reliable path to clinical success.
Cell therapy manufacturing involves the complex process of developing, scaling, and producing living cell–based treatments. They are addressing various diseases such as cancer, genetic disorders, and autoimmune conditions. Unlike traditional pharmaceuticals, cell therapies require individualized, high-precision workflows that preserve cell viability and function.
Cell therapies can either be autologous or allogeneic:
Autologous cell therapies: Autologous cell therapies involve collecting a patient’s own cells, modifying or expanding them outside the body, and then reinfusing them to repair, regenerate, or fight diseases. As these cells originate from the same individual, the risk of immune rejection is reduced compared to donor-based therapies.
Allogeneic cell therapies: Allogeneic cell therapies use cells donated from a healthy individual, which are processed and administered to a different patient to treat disease or restore function. Unlike autologous approaches, they can be produced in larger batches as “off-the-shelf” treatments but carry a higher risk of immune rejection.
The manufacturing processes must comply with stringent cGMP standards and ensure safety, efficacy, and reproducibility.
From lab to clinical production, the cell therapy manufacturing process involves multiple interdependent stages:
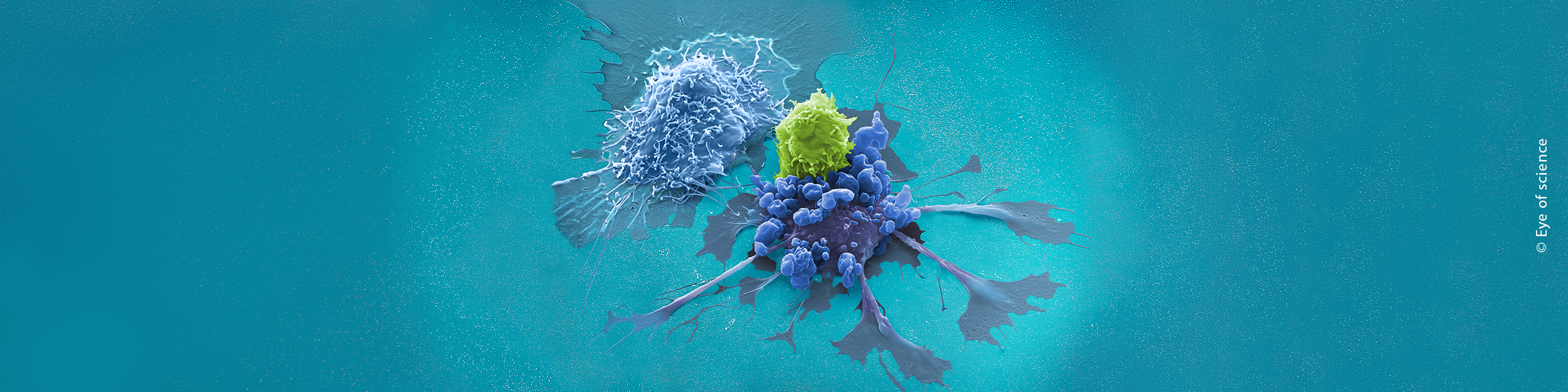
Today, several cell and gene therapies have transformed patient treatment, with CAR T cell therapy emerging as a particularly notable success. The manufacturing process follows a carefully orchestrated sequence. Immune cells are first collected from either the patient or a donor. T cells are then isolated and genetically modified to express specialized receptors – Chimeric Antigen Receptors (CARs) – designed to recognize and bind to cancer cells. Following modification, these engineered CAR T cells are expanded in culture before being infused back into the patient. Once reintroduced into the body, they actively seek out and eliminate cancer cells, delivering a highly personalized and potent therapeutic approach.
Although the cell and gene therapy field has made significant progress in recent years, the manufacturing process remains highly challenging. From isolating cells from patients or donors to performing complex cell engineering, manufacturers face difficult-to-scale operations characterized by high costs, labor-intensive manual production, and lack of standardization in quality control and validation.

Miltenyi Bioindustry addresses these manufacturing challenges through automated, closed-system processes and proven platform technologies that improve scalability, reduce variability, and accelerate development timelines from concept to commercialization.
Miltenyi Bioindustry enables scalable manufacturing with the closed, automated CliniMACS Prodigy Platform and the CliniMACS Cell Factory® ballroom concept for clinical and commercial production.
Miltenyi Bioindustry is on your side from start to finish
All services are built around speed, precision, and compliance.
With five manufacturing centers strategically located worldwide, we ensure proximity, regulatory alignment, and production flexibility.
These facilities meet stringent GMP standards and are equipped with modular cleanroom infrastructures, automated production suites, and scalable vector manufacturing capabilities.
We distinguish ourselves with three key advantages:
Platforms, standards, infrastructure
Our proprietary CliniMACS Prodigy and MACS Quant® Cell Analyzer systems enable automation, standardization, and traceability across processes.
People: Expertise across all phases
Our cross-functional teams bring decades of experience in cell therapy development, scale-up, and regulatory pathways.
IND readiness acceleration
We help reduce time-to-IND with strategic development frameworks, risk mitigation tools, and integrated regulatory guidance.
What is cell therapy manufacturing?
It is a highly controlled process where living cells from patients or donors are isolated, modified, expanded, and formulated into a therapeutic product.
What are the raw materials for cell therapy?
Starting materials typically include patient or donor cells, growth media, cell activation matrix, viral vectors, cytokines, and buffers.
What are the steps in cell therapy manufacturing?
Cell collection -> Engineering -> Expansion -> Harvest and formulation
What are the steps for scaling up a cell therapy process to GMP manufacturing?
Automated and closed systems, such as the CliniMACS Prodigy Platform, provide scalable options by simplifying technology transfer and enabling use across multiple instruments.
What’s the difference between autologous and allogeneic manufacturing?
Autologous therapies use the patient’s own cells, while allogeneic therapies use donor cells that can treat multiple patients.
What are the cleanroom standards for cell therapy GMP?
ISO-classified cleanrooms, strict air quality control, and aseptic procedures are required for cGMP compliance.
Which vectors are most common for gene delivery?
Vesicular Stomatitis Virus glycoprotein (VSV-G ) and baboon envelope pseudotyped (BaEV) lentiviral vectors are among the most widely used for gene-modified cell therapies.
For more information, detailed service brochures, or partnership inquiries, please contact us directly or explore our service pages.